Several batches of Doliprane are being recalled due to a risk of overdose and potentially liver damage. What batches are affected? What to do if you have them at home?
It is an essential product in the medicine cabinet which is the subject of special attention today. The National Medicines Safety Agency (ANSM) is recalling 27 batches of Doliprane 2.4% (paracetamol) on Monday February 23 due to a defect which could lead to a dosage error. This is the well-known pink oral suspension used to relieve pain and fever in infants and children weighing 3 to 26 kg.
The Medicines Agency, as well as the Opella Healthcare France laboratory which markets it, specifies that the quality of the syrup itself is not called into question. The problem comes from the accessory: the graduated pipette. Due to a manufacturing issue at one of the production sites, the graduations on some pipettes gradually fade after being rinsed with warm or hot water. Without legible graduations, it becomes impossible to precisely measure the dose of paracetamol to be administered. This can lead to an overdose, including liver damage. “This risk, although low, exists in babies and young children in whom liver damage may occur if repeated doses are too high.“, specifies the ANSM in its press release.
As a precaution and to ensure patient safety, the recall concerns boxes distributed in pharmacies between November 12, 2025 and February 3, 2026. Here is the complete list of the 27 lots:
- L033 (EXP 08/2027);
- L034 (EXP 08/2027);
- L035 (EXP 08/2027);
- L036 (EXP 08/2027);
- L037 (EXP 09/2027);
- L038 (EXP 09/2027);
- L039 (EXP 09/2027);
- L040 (EXP 09/2027);
- L041 (EXP 09/2027);
- L042 (EXP 09/2027);
- L043 (EXP 09/2027);
- L044 (EXP 09/2027);
- L045 (EXP 09/2027);
- L046 EXP (09/2027);
- L047 (EXP 09/2027);
- L048 (EXP 09/2027);
- L049 (EXP 09/2027);
- L050 (EXP 10/2027);
- L051 (EXP 10/2027);
- L053 (EXP 10/2027);
- L054 (EXP 10/2027);
- L055 (EXP 10/2027);
- L056 (EXP 10/2027);
- L057 (EXP 10/2027);
- L058 (EXP 10/2027);
- L061 (EXP 10/2027);
- L062 (EXP 10/2027).
To find the batch number, simply look on the back of the medicine box, in the white box where the expiration date appears, or directly on the bottle label (see photo below). If your lot number is not listed, your medication is not affected by the recall. On the other hand, if you have one of the bottles mentioned above, here is what to do: immediately stop using the pipette provided in the box. Do not replace it with the pipette of another medication (dosages vary depending on the product).
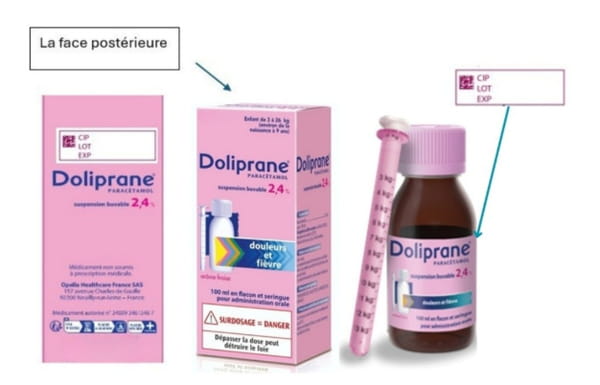
Return the entire box (bottle + pipette) to your pharmacist. A free exchange will be offered to you for a compliant box. No panic for future purchases, the laboratory has clarified that stocks are sufficient to ensure the availability of the product in pharmacies. The defect was identified and corrected on the new production lines.
To date, no adverse effects related to this defect have been reported. However, vigilance remains essential. If you are concerned that you have administered too high a dose or if your child has unusual symptoms (nausea, vomiting, paleness, abdominal pain), contact your doctor or pediatric emergency room promptly.









